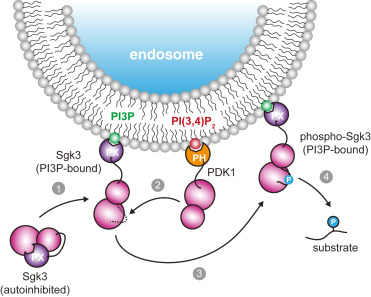
How the highly curved phagophore membrane is stabilized during autophagy initiation is a major open question in autophagosome biogenesis. Here, we use in vitro reconstitution on membrane nanotubes and molecular dynamics simulations to investigate how core autophagy proteins in the LC3 (Microtubule-associated proteins 1A/1B light chain 3) lipidation cascade interact with curved membranes, providing insight into their possible roles in regulating membrane shape during autophagosome biogenesis. ATG12(Autophagy-related 12)–ATG5-ATG16L1 was up to 100-fold enriched on highly curved nanotubes relative to flat membranes. At high surface density, ATG12–ATG5-ATG16L1 binding increased the curvature of the nanotubes. While WIPI2 (WD repeat domain phosphoinositide-interacting protein 2) binding directs membrane recruitment, the amphipathic helix α2 of ATG16L1 is responsible for curvature sensitivity. Molecular dynamics simulations revealed that helix α2 of ATG16L1 inserts shallowly into the membrane, explaining its curvature-sensitive binding to the membrane. These observations show how the binding of the ATG12–ATG5-ATG16L1 complex to the early phagophore rim could stabilize membrane curvature and facilitate autophagosome growth.
J Biol Chem 297(2) 100919
D. Pokorny, L. Truebestein, K. D. Fleming, J. E. Burke, and T. A. Leonard
J Biol Chem 297(2) 100919
D. Pokorny, L. Truebestein, K. D. Fleming, J. E. Burke, and T. A. Leonard