The rapid and dynamic activation of the innate immune system is achieved through complex signaling networks regulated by post-translational modifications modulating the subcellular localization, activity, and abundance of signaling molecules. Many constitutively expressed signaling molecules are present in the cell in inactive forms, and become functionally activated once they are modified with ubiquitin, and, in turn, inactivated by removal of the same post-translational mark. Moreover, upon infection resolution a rapid remodeling of the proteome needs to occur, ensuring the removal of induced response proteins to prevent hyperactivation. This review discusses the current knowledge on the negative regulation of innate immune signaling pathways by deubiquitinating enzymes, and through degradative ubiquitination. It focusses on spatiotemporal regulation of deubiquitinase and E3 ligase activities, mechanisms for re-establishing proteostasis, and degradation through immune-specific feedback mechanisms vs. general protein quality control pathways.
J Biol Chem 297(2) 100919
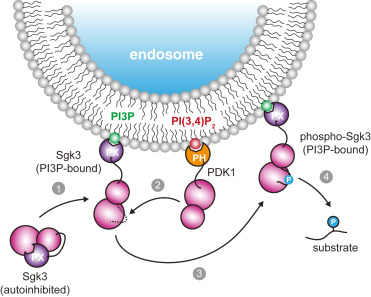
D. Pokorny, L. Truebestein, K. D. Fleming, J. E. Burke, and T. A. Leonard
J Biol Chem 297(2) 100919
D. Pokorny, L. Truebestein, K. D. Fleming, J. E. Burke, and T. A. Leonard